Ionic EquilibriumHard
Question
A solution containing a mixture of 0.05 M NaCl and 0.05 M NaI is taken (Ksp of AgCl = 10-10 and Ksp of AgI = 4 × 10-16). When AgNO3 is added to such a solution.
Options
A.the concentration of Ag+ required to precipitate Cl- = 2 ×10-9 mol/L.
B.the concentration of Ag+ required to precipitate I- = 8 × 10-15 mol/L.
C.AgCl and AgI will be precipitated together.
D.first AgI will be precipitated.
Solution
[Ag+] ion required to precipitate Cl- ion = 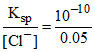 = 2 × 10-9 mol/L.
= 2 × 10-9 mol/L.
[Ag+] ion required to precipitate I- ion =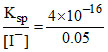 = 8 × 10-15 mol/L.
= 8 × 10-15 mol/L.
Since [Ag+] ion conc. required to precipitate AgI is less than the [Ag+] ion conc. required to precipitate AgCl, AgI precipitates first.
Hence choices (A),(B) and (D) are correct while (C) is incorrect.
[Ag+] ion required to precipitate I- ion =
Since [Ag+] ion conc. required to precipitate AgI is less than the [Ag+] ion conc. required to precipitate AgCl, AgI precipitates first.
Hence choices (A),(B) and (D) are correct while (C) is incorrect.
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
25 ml of a solution of barrium hydroxide on titration with a 0.1 molar solution of hydrochloric acid gave a litre value ...1 M NaCl and 1 M HCl are present in an aqueous solution. The solution is...The solubility product of Co(OH)3 is 2.7 × 10−43. The pH of saturated solution of Co(OH)3 is about...The pKa of a weak acid, HA, is 4.80. The pKb of a weak base, BOH, is 4.78. The pH of an aqueous solution of the correspo...pOH of H2O is 7.0 at 298 K. If water is heated at 350 K, which of the following statement should be true?...