Ionic EquilibriumHard
Question
2.5 mL of 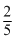 M weak monoacidic base(Kb = 1 × 10-12 at 25o C) is titrated with
M weak monoacidic base(Kb = 1 × 10-12 at 25o C) is titrated with 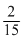 M HCl in water at 25oC. The concentration of H+ at equivalence point is (Kw = 1 × 10 -14 at 25oC)
M HCl in water at 25oC. The concentration of H+ at equivalence point is (Kw = 1 × 10 -14 at 25oC)
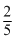 M weak monoacidic base(Kb = 1 × 10-12 at 25o C) is titrated with
M weak monoacidic base(Kb = 1 × 10-12 at 25o C) is titrated with 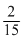 M HCl in water at 25oC. The concentration of H+ at equivalence point is (Kw = 1 × 10 -14 at 25oC)
M HCl in water at 25oC. The concentration of H+ at equivalence point is (Kw = 1 × 10 -14 at 25oC)Options
A.3.7 × 10-13 M
B.3.2 × 10-7 M
C.3.2 × 10-2 M
D.2.7 × 10-2 M
Solution
BOH + HCl → BCl + H2O
C
B+ H2O ⇋ BOH + H+
C(1 - h) Ch Ch
Volume of HCl used =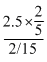 = 7.5ml
= 7.5ml
Concentration of Salt, C =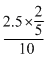 = 0.1 M
= 0.1 M
∴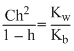
Solving h = 0.27
[H+] = Ch = 0.1 × 0.27 = 2.7 × 10-2 M
C
B+ H2O ⇋ BOH + H+
C(1 - h) Ch Ch
Volume of HCl used =
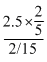 = 7.5ml
= 7.5mlConcentration of Salt, C =
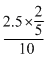 = 0.1 M
= 0.1 M∴
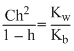
Solving h = 0.27
[H+] = Ch = 0.1 × 0.27 = 2.7 × 10-2 M
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
An example of a reversible reaction is :...The solubility products of Mg(OH)2, Cd(OH)2, Al(OH)3 and Zn(OH)2 are 4 × 10–11, 8 × 10–6, 8.5 × 10–23 and 1.8 × 10–14, r...Which one is the correct graph (figure) for the corresponding acid base titration?...pH value of which of the following is not equal to one :...At 90o C, the hydronium ion concentration in pure water is 10−6 M. If 100 ml of 0.5 M-NaOH solution is mixed with 250 ml...