Ionic EquilibriumHard
Question
Two buffer solutions, A and B, each made with acetic acid and sodium acetate differ in their pH by one unit, A has salt : acid = x : y, B has salt : acid = y : x. If x > y, then the value of x : y is
Options
A.10,000
B.3.17
C.6.61
D.2.10
Solution
Buffer (A):
pH1 = pKa + log(x/y) .... (i)
Buffer (B):
pH2 = pKa + log (y/x) ..... (ii)
Since x > y.
∴ pH1 - pH2 = 1 = log (x/y) - log (y/x)
∴ 1 = 2 log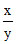
log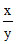 =
= 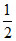 = 0.5
= 0.5
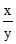 = Antilog (0.5) = 3.17.
= Antilog (0.5) = 3.17.
pH1 = pKa + log(x/y) .... (i)
Buffer (B):
pH2 = pKa + log (y/x) ..... (ii)
Since x > y.
∴ pH1 - pH2 = 1 = log (x/y) - log (y/x)
∴ 1 = 2 log
log
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
In a buffer solution containing equal concentration of B- and HB, the Kb for B-1 is 10-10 . The pH of buffer solution is...To prepare a buffer of pH 8.26 amount of (NH4)2 SO4 to be added to 500 mL of 0.01 M NH4OH solution [pKa (NH4+) = 9.26] i...Complete hydrolysis of cellulose gives...Which of the following has the highest degree of ionisation ?...The solubility product of BaCrO4 is 2.4 × 10-10 M2. The maximum concentration of Ba(NO3)2 possible without precipit...