Ionic EquilibriumHard
Question
Complete hydrolysis of cellulose gives
Options
A.D-ribose
B.D-glucose
C.L-glucose
D.D-fructose
Solution
(C6H10O5)n + nH2O 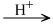 nC6H12O6
nC6H12O6
D - Glucose
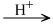 nC6H12O6
nC6H12O6D - Glucose
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Solubility of BaF2 in a solution of Ba(NO3)2 will be represented by the concentration term:...A solution having hydrogen ion concentration is 0.0005 g eqvt./litre, its pOH is :...The pH value of ordinary water is :...The solubility of Ag2CO3 in water at 25oC is 1 × 10-4 mole/litre. What is its solubility in 0.01 M Na2CO3 solution?...The pH of the solution obtanied by mixing 10 mL of 10-1 N HCI and 10 mL of 10-1 N NaOH is:...