Ionic EquilibriumHard
Question
In a buffer solution containing equal concentration of B- and HB, the Kb for B-1 is 10-10 . The pH of buffer solution is
Options
A.10
B.7
C.6
D.4
Solution
(i) For basic buffer
pOH = pKb + log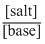
(ii) pH + pOH = 14
Given, Kb = 1 × 10-10, [ salt ] = [ base ]
pOH = - log Kb + log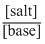
∴ pOH = - log ( 1 × 10-10) + log1 = 10
pH + pOH = 14
pH = 14 - 10 = 4
pOH = pKb + log
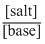
(ii) pH + pOH = 14
Given, Kb = 1 × 10-10, [ salt ] = [ base ]
pOH = - log Kb + log
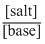
∴ pOH = - log ( 1 × 10-10) + log1 = 10
pH + pOH = 14
pH = 14 - 10 = 4
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
0.1 millimole of CdSO4 are present in 10 mL acid solution of 0.08 N HCl. Now H2S is passed to precipitate all the Cd2+ i...A 40.0 ml solution of weak base, BOH is titrated with 0.1 N – HCl solution. The pH of the solution is found to be 10.0 a...Equimolar solutions of the following substances were prepared separately. Which one of these will record the highest pH ...The pKa of acetylasliclic acid (aspirin) is 3.5. The pH of gastric juice in human stomach is about 2-3 and the pH in the...In which of the following cases, pH is greater than 7 ?...