Ionic EquilibriumHard
Question
To prepare a buffer of pH 8.26 amount of (NH4)2 SO4 to be added to 500 mL of 0.01 M NH4OH solution [pKa (NH4+) = 9.26] is
Options
A.0.05 mole
B.0.025 mole
C.0.10 mole
D.0.005 mole
Solution
For the buffer solution of NH3 & NH4+
pH = pKa + log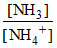 ⇒ 8.26 = 9.26 + log
⇒ 8.26 = 9.26 + log 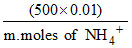
⇒ m. moles of NH4+ = 50 ∴ moles of (NH4)2 SO4 required = 0.025.
pH = pKa + log
⇒ m. moles of NH4+ = 50 ∴ moles of (NH4)2 SO4 required = 0.025.
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Which statement is/are correct:...Which of the following is the strongest base :-...In which of the following combinations, is buffer action expected?1. NH3 + NH4CI in 1 : 1 mole ratio2. HCI + NaCI in 1 :...The solubility of Ag2CO3 in water at 25oC is 1 × 10-4 mole/litre. What is its solubility in 0.01 M Na2CO3 solution?...The pH of which salt is independent of its concentration :1. (CH3COO)C5H5NH 2. NaH2PO4 3. Na2HPO4 4.NH4CN...