Heat and Thermal ExpansionHard
Question
The density in grams per litre of ethylene (C2H4) at STP is : -
Options
A.1.25
B.2.50
C.3.75
D.4.25
Solution
PV = nRT ⇒ P = 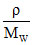 RT
RT
⇒ ρ =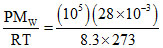 kg m-3 = 1.25 g/litre
kg m-3 = 1.25 g/litre
⇒ ρ =
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
A system S receives heat continuously from an electrical heater of water 10W. The temperature of S becomes constant at 5...An ideal monotonic gas undergoes a cycle ABCA as shown in the fig. The ratio of heat absorbed during AB to the work done...One mole of an ideal gas ungderoes a whose molar heat capacity is 4R and in which work done by gas for small change in t...A inert gas obeys the law PVx = constant. For what value of x, it has negative molar specific heat -...In the following figure a wire of cross sectional radius r is fixed lightly at P and Q. The Young′s modulus of the...