Heat and Thermal ExpansionHard
Question
One mole of an ideal gas ungderoes a whose molar heat capacity is 4R and in which work done by gas for small change in temperature is given by the relation dW = 2RdT, then the ration 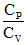 is.
is.
Options
A.7/5
B.5/3
C.3/2
D.2
Solution
For small change dQ = dU + dW
nCdT = nCVdT + 2nRdT ∴ C = CV + 2R ; 4R = CV + 2R ∴ CV = 2R
Allso CV =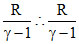 = 2R ⇒ 2γ - 2 = 1 ⇒ γ = 32
= 2R ⇒ 2γ - 2 = 1 ⇒ γ = 32
nCdT = nCVdT + 2nRdT ∴ C = CV + 2R ; 4R = CV + 2R ∴ CV = 2R
Allso CV =
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
The expansion of unit mass of a constant pressure is shown in the diagram. here : -...If water at 0oC , kept in a container with an open top, is placed in a large evacuated chamber :-...A thermocouple is made from two metals, Antimony and Bismuth. If one junction of the couple is kept hot and the other is...5n, n and 5n moles of a monoatomic, diatomic and non - linear polyatomic gases (which do not react chemically with each ...The figure shows two paths for the change of state of a gas from A to B. The ratio of molar heat capacities in path 1 an...