Heat and Thermal ExpansionHard
Question
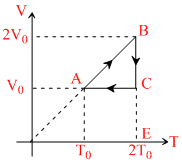
An ideal monotonic gas undergoes a cycle ABCA as shown in the fig. The ratio of heat absorbed during AB to the work done on the gas during BC is : -

Options
A.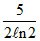
B.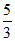
C.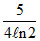
D.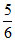
Solution
WAB = (2V0 - V0)P0 = P0V0
[isobaric process]
WBC =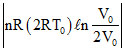 = 2P0v0l N2
= 2P0v0l N2
[Isothermal process]
∴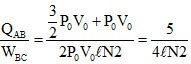
[isobaric process]
WBC =
[Isothermal process]
∴
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
Three rod of some dimensions are arranged as shown in the figure. They have thermal conductivities k1, k2 & k3. The poin...A wooden wheel of radius R is made of twosemicircular parts (see figure). The two partsare held together by a ring made ...A thermally insulated vessel contains some water at 0oC. The vessel connected to a vacuum pump out water vapor. This res...A steel scale is to be prepared such that the millimeter intervals are to be accurate4 within 6 × 10-5 mm.The maxim...The internal energy of as gas is given U = 5 + 2PV. It expands from V0 to 2V0 against a constant pressure P0. The heat a...