Heat and Thermal ExpansionHard
Question
A system S receives heat continuously from an electrical heater of water 10W. The temperature of S becomes constant at 50oC when the surrounding temperature is 20oC After the heater is switch off, S cools from 35.1oC to 34.9oC in 1 minuteThe heal capacity S is: -
Options
A.100 J / oC
B.300 J / oC
C.750 J / oC
D.1500 J / oC
Solution
Rate of cooling
= ms
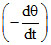 = 4σAT03ᐃT
= 4σAT03ᐃT
⇒ 4σA × T03ᐃ (50 - 20) = 10
and 4σA × T03ᐃ (35 - 20) = ms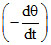 = ms
= ms 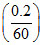
⇒ ms =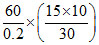 = 1500 J / oC
= 1500 J / oC
= ms
⇒ 4σA × T03ᐃ (50 - 20) = 10
and 4σA × T03ᐃ (35 - 20) = ms
⇒ ms =
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
In a 20 m deep lake, the bottom is at a constant temperature of 4oC. the air temperature is constant at -10oC. The therm...Water contained in a jar at room temperature (20oC) is intended to be colled by method - I or method - II given below :M...A gas based geyser heats water flowing at the rate of 5.0 litres per minute from $27^{\circ}C$ to $87^{\circ}C$.The rate...Certain prefect gas is found to obey PVn = constant during adiabatic process. The volume expansion cofficient at tempera...One end of a horizontal thick copper wire of length 2L and radius 2R is welded to an end of another horizontal thin copp...