Chemical Kinetics and Nuclear ChemistryHard
Question
Read the following industrial methods for the preparation of H2SO4 and answer the question at the end. Professor Molina of the Massachusetts Institute of Technology won the 1995 Nobel Prize in Chemistry for his work on atmospheric chemistry. One reaction that he has studied in detail is the acid rain reaction which produces H2SO4 in the atmosphere. He has proposed two possible stoichiometric reactions :
Proposal A : H2O (g) + SO3(g) → H2SO4(g)
Proposal B : 2 H2O (g) + SO3(g) → H2SO4 (g) + H2O (g)
Using simple collision theory, what reaction orders would be expected for proposal B ?
Proposal B is thought to proceed by the following two-step process :
SO3 + 2 H2O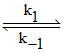 SO3.2 H2O (fast)
SO3.2 H2O (fast)
SO3.2 H2O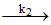 H2SO4 + H2O (slow)
H2SO4 + H2O (slow)
(SO3.2 H2O is a complex which is stabilized by hydrogen bonds and k2 << k1 or k-1).
Proposal A : H2O (g) + SO3(g) → H2SO4(g)
Proposal B : 2 H2O (g) + SO3(g) → H2SO4 (g) + H2O (g)
Using simple collision theory, what reaction orders would be expected for proposal B ?
Proposal B is thought to proceed by the following two-step process :
SO3 + 2 H2O
SO3.2 H2O
(SO3.2 H2O is a complex which is stabilized by hydrogen bonds and k2 << k1 or k-1).
Options
A.k [H2O] [SO3]
B.k[H2O]2 [SO3]
C.k [SO3]
D.k[H2O]
Solution
Rate determining step is slow step
Then Rate = k2 [SO3 . 2 H2O] .....(i)
We know by fast equation
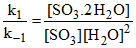
[SO3. 2 H2O] =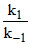 [SO3] [H2O]2 ....(ii)
[SO3] [H2O]2 ....(ii)
put the value of [SO3 . 2 H2O] from (ii) to (i)
Rate = k2 ×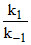 [SO3 ] [ H2O]2
[SO3 ] [ H2O]2
Rate = k [H2O]2 [SO3]
Then Rate = k2 [SO3 . 2 H2O] .....(i)
We know by fast equation
[SO3. 2 H2O] =
put the value of [SO3 . 2 H2O] from (ii) to (i)
Rate = k2 ×
Rate = k [H2O]2 [SO3]
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
As the initial concentration increases from 0.75 to 1.55 M in a reaction, t1/2 decreases from 60 to 29 s. The order of t...T50 (Half-life period) of first-order reaction is 10 minute. Starting with 10 mol L-1, rate after 20 minute is :...For the non-equilibrium process, A + B → products, the rate is first-order w.r.t. A and second-order w.r.t. B. If ...The time taken in 75% completion of a zero-order reaction is 10 h. In what time, the reaction will be 90% completed?...What is ᐃH for the reaction A + B → C, where the mechanism involves several kinetic steps....