Mole ConceptHard
Question
One of N2 is mixed with 3 mole of H2 in a closed vessel of 3 ltr. 20% of N2 is converted into NH3 then Kc for
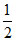 N2 +
N2 + 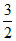 H2 ⇋ NH3 is :
H2 ⇋ NH3 is :
Options
A.0.36 lit/mole
B.0.46 lit/mole
C.0.50 lit/mole
D.0.2 lit/mole
More Mole Concept Questions
The weight of one molecule of a compound of molecular formula C60H122 is...The largest number of molecules is in :...The solubility of Mg(OH)2 is S moles/litre. The solubility product under the same condition is...The strength of 10-2 M Na2CO3 solution in terms of molality will be (density of solution = 1.10 g mL-1). (Molecular weig...pH of a saturated solution of Ba ( OH )2 is 12. The value of solubility product Ksp of Ba ( OH2 ) is...