Mole ConceptHard
Question
The largest number of molecules is in :
Options
A.36 g of water
B.28 g of caebon monoxide
C.46 g of ethyl alcohol
D.54 g of nitrogen pentoxide
Solution
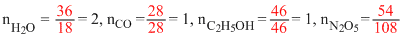
∵ nH2 is largest, so maximum number of molecules are present in 36 g H2O
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
Which of the following is not correctly matched :-...If the mass of neutron is doubled and that of proton is halved, the molecular mass of H2O containing only H1 and O16 ato...How many moles of electron weigh one kilogram :...6.02 × 1020 molecules of urea are present in 100 ml of its solution. The concentration of urea solution is...If NA is Avogadro′s number then number of valence electrons in 4.2 g of nitride ions (N3-)...