Mole ConceptHard
Question
The strength of 10-2 M Na2CO3 solution in terms of molality will be (density of solution = 1.10 g mL-1). (Molecular weight of Na2CO3 = 106 g mol-1)
Options
A.9.00 × 10-3
B.1.5 × 10-2
C.5.1 × 10-3
D.11.2 × 10-3
Solution
(A) Explanation : m = 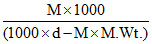 where ′m′ is molality, M is molarity.
where ′m′ is molality, M is molarity.
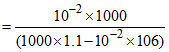
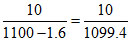 = 9.00 × 10-3
= 9.00 × 10-3
[Take 1099.4 = 1100]
[Take 1099.4 = 1100]
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
Volume of a gas at NTP is 1.12 × 10-7cc. The number of molecule in it is :...An amount of 2 moles of KClO3 is decomposed completely to produce O2 gas. How many moles of butene C4H8 can be burnt com...The mass of 3.2 × 105 atoms of an element is 8.0 × 10−18 g. The atomic mass of the element is about(NA = 6 × 1023)...2KI + I2 + 22 HNO3 → 2HIO3 + 2KIO3 + 22NO2 + 10H2OIf 3 mole of KI & 2 moles I2 are reacted with excess of HNO3. Vo...To determine soluble (free) SiO2 in a rock, an alkaline extraction was carried out, as a result of which there was found...