Mole ConceptHard
Question
pH of a saturated solution of Ba ( OH )2 is 12. The value of solubility product Ksp of Ba ( OH2 ) is
Options
A.3.3 × 10-7
B.5.0 × 10-7
C.4.0 × 10-6
D.5.0 × 10-6
Solution
Given, pH of Ba ( OH )2 = 12
∴ pOH = 14 - pH
= 14 - 12 = 2
We know that,
pOH = - log [ OH- ]
2 = - log [ OH- ]
[ OH- ] = antilog ( -2 )
[ OH- ] = 1 × 10-2
Ba ( OH )2 dissolves in water as
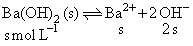
∴ [ OH- ] = 2s = 1 × 10 -2
[ Ba2+ ]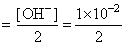
Ksp = [ Ba2+ ] [ OH- ]2
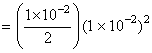
= 0.5 × 10-6 = 5 × 10-7
∴ pOH = 14 - pH
= 14 - 12 = 2
We know that,
pOH = - log [ OH- ]
2 = - log [ OH- ]
[ OH- ] = antilog ( -2 )
[ OH- ] = 1 × 10-2
Ba ( OH )2 dissolves in water as
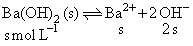
∴ [ OH- ] = 2s = 1 × 10 -2
[ Ba2+ ]
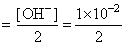
Ksp = [ Ba2+ ] [ OH- ]2
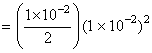
= 0.5 × 10-6 = 5 × 10-7
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
Consider the following statements1. If all the reactants are not taken in their stoichiometric ratio, then at least one ...Dissolving 120 g of urea (mol. wt. 60) in 1000 g of water gave a solution of density 1.15 g/mL. The molarity of the solu...Vapour pressure of a liquid solution increases -...When 22.4 litres of H2(g) is mixed with 11.2 liters of Cl2 (g), each at STP, the moles of HCl(g) formed is equal to...Change in volume of the system does not alter the number of moles in which of the following equilibria ?...