Mole ConceptHard
Question
The weight of one molecule of a compound of molecular formula C60H122 is
Options
A.1.2 × 10-20 g
B.5.025 × 1023 g
C.1.4 × 10-21 g
D.6.023 × 10-20 g
Solution
M.W. = 60 × 12 + 122 = 842
Weight of one molecule =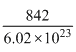 gm
gm
= 140 × 10-23 gm = 1.4 × 10-21 gm
Weight of one molecule =
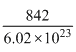 gm
gm= 140 × 10-23 gm = 1.4 × 10-21 gm
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
From 2 mg calcium, 1.2 × 1019 atoms are removed. The number of g-atoms of calcium left is (Ca = 40)...Vapour density of a volatile substance is 1.2 (C2H6 = 1). Its molecular mass would be...The energy of activation is :-...V1 ml of unknown gas (A) + V2 ml of O2 → (V1 + V2) ml of CO2. Gas ‘A’ may be...The distribution of molecular kinetic energy at two temperatures is as shown in the following graph:Which of the followi...