Chemical EquilibriumHard
Question
Calculate internal energy charge in the following equation at 100oC ?
H2 +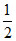 O2 → H2O + 68.4 Kcal
O2 → H2O + 68.4 Kcal
H2 +
Options
A.68.4 Kcal
B.- 68.4 Kcal
C.- 68.03 Kcal
D.none of these
More Chemical Equilibrium Questions
The equilibrium constant for the reaction 3C2H2 $\rightleftharpoons$C6H6 is 4.0 at T K. If the equilibrium concentration...The Deacon reaction is the oxidation of HCl by O2 in the following reactionHCl(g) + ¼ O2(g) $\rightleftharpoons$½ Cl2(g)...The formation constant of Ni(NH3)62+ is 6 × 108 at 250C. If 50 ml of 2.0 M NH3 is added to 50 ml of 0.20 M solution...For the equilibrium SrCl2 · 6H2O(s) ⇋ SrCl2 · 2H2O(s) + 4H2O (g) the equilibrium constant KP = 16 × 10-12 atm...The amounts of 0.8 mol of PCl5 and 0.2 mole of PCl3 are mixed in a 1 L flask. At equilibrium, 0.4 mole of PCl3 is presen...