Chemical EquilibriumHard
Question
The formation constant of Ni(NH3)62+ is 6 × 108 at 250C. If 50 ml of 2.0 M NH3 is added to 50 ml of 0.20 M solution of Ni2+, the concentration of Ni2+ ion will be nearly equal to :
Options
A.3 × 10-10 mole litre-1
B.2 × 10-10 mole litre-1
C.2 × 10-9 mole litre-1
D.4 × 10-8 mole litre-1
Solution
Ni+2 + 6NH3 → [Ni (NH3)]+6 Kf = 6 × 108
t = 0 0.01 mole 0.1 mole 0
Kc =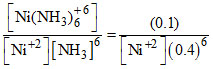 = 6 × 108. [Ni+2] = 4 × 10-8.
= 6 × 108. [Ni+2] = 4 × 10-8.
t = 0 0.01 mole 0.1 mole 0
Kc =
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
In respect of the equation k = Ae-Ea/RT in chemical kinetics, which one of the following statements is correct?...At a certain temperature, the value of KP for the reaction: 2CO(g) $\rightleftharpoons$CO2(g) + C(graphite) is 0.1 atm−1...The following equilibria are given : N2 + 3H2 ⇋ 2NH3 K1 N2 + O2 ⇋ 2NO K2 H2 + O2 ⇋ H2O K3 The equilibr...1 mole each of H2(g) and I2(g) are introduced in a 1L evacuated vessel at 523K and equilibrium H2(g) + I2(g) ⇋ 2HI...A vessel at 1000 K contains CO2 with a pressure of 0.5 atm. Some of the CO2 is converted into CO on the addition of grap...