Chemical EquilibriumHard
Question
For the equilibrium SrCl2 · 6H2O(s) ⇋ SrCl2 · 2H2O(s) + 4H2O (g) the equilibrium constant KP = 16 × 10-12 atm4 at 1oC. If one litre of air saturated with water vapour at 1oC is exposed to a large quantity of SrCl2· 2H2O(s), what weight of water vapour will be absorbed? Saturated vapour pressure of water at 1oC = 7.6 torr.
Options
A.6.4 mg
B.3.25 mg
C.2.3 mg
D.8.5 mg
Solution
SrCI2 . 6H2O(s) ⇋ SrCI2 . 2H2O(s) + 4H2O(g) Kp = 16 × 10-12
(PH2O)4 = Kp PH2O = (KP)1/4 = 2 × 10-3 atm
H2O (l) ⇋ H2O(g) pH2O =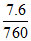 = 1.0 × 10-2
= 1.0 × 10-2
nH2O =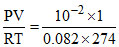 = 4.45 × 10-4
= 4.45 × 10-4
nH2O =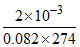 = 8.9 × 10-5 ∴ nH2O = absorbed = 3.56 × 10-4
= 8.9 × 10-5 ∴ nH2O = absorbed = 3.56 × 10-4
∴ wt absorbed = 6.4 mg. Therefore, (A) option is correct.
(PH2O)4 = Kp PH2O = (KP)1/4 = 2 × 10-3 atm
H2O (l) ⇋ H2O(g) pH2O =
nH2O =
nH2O =
∴ wt absorbed = 6.4 mg. Therefore, (A) option is correct.
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
If for 2A2B(g) ⇋ 2A2(g) + B2(g), Kp = TOTAL PRESSURE (at equilibrium) and starting the dissociation from 4 mol of ...The complexion of Fe2+ with the chelating agent dipyridyl has been studied kinetically in both the forward and reverse d...For a gaseous equilibrium 2A(g) $\rightleftharpoons$2B(g) + C(g), KP has a value 1.8 at 700 K. The value of KC for the e...The equilibrium constant for some reactions are given below against each of the reaction.(i) 2N2 + 5O2$\rightleftharpoon...The correct relationship between free energy change in a reaction and the corresponding equilibrium constant Kc is...