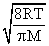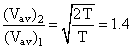Gaseous StateHard
Question
By what factor does the average velocity of a gaseous molecule increase when the temperature (in Kelvin) is doubled ?
Options
A.2.8
B.4.0
C.1.4
D.2.0
More Gaseous State Questions
Maximum deviation from ideal gas is expected from :...If the pressure of a gas contained in a closed vessel is increased by 0.4 % when heated by 1oC its initial temperature m...A real gas most closely approaches the behaviour of an ideal gas at -...When CO2 under high pressure is released from a fire extinguisher, particles of solid CO2 are formed, despite the low su...X ml of H2 gas effuses through a hole in a container in 5 sec. The time taken for the effusion of the same volume of the...