Gaseous StateHard
Question
Density of a Vander Waal′s gas at 500 K and 1 atm is found to be 0.8 kg/m3. It is also found that the gas diffuses 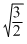 times slower than pure O2 gas under identical conditions. The value of Z for the real gas is (R = 0.08 atm - l /mol-K) :
times slower than pure O2 gas under identical conditions. The value of Z for the real gas is (R = 0.08 atm - l /mol-K) :
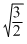 times slower than pure O2 gas under identical conditions. The value of Z for the real gas is (R = 0.08 atm - l /mol-K) :
times slower than pure O2 gas under identical conditions. The value of Z for the real gas is (R = 0.08 atm - l /mol-K) :Options
A.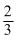
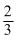
B.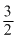
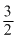
C.1.8
D.0.8
Solution
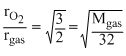 ⇒ Mgas = 48
⇒ Mgas = 48Vm, real =
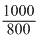 × 48 l = 60l
× 48 l = 60lVm, ideal =
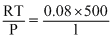 = 40 l
= 40 lZ =
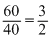
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
At constant pressure which of the following does not represent Charle′s law ?...If the pressure of a gas contained in a closed vessel is increased by 0.4 % when heated by 1oC its initial temperature m...For real gases van der Waals equation is written as (V - nb) = nRT where ′a′ and ′b′ are van der...Gas Y + B → milkiness,Gas ′X′ burn with Blue flame then correct choice is :-...A gaseous mixture containing He, CH4 & SO2 was allowed to effuse through a fine hole then find what molar ratio of gases...