Gaseous StateHard
Question
At 100oC and 1 atm if the density of the liquid water is 1.0 g cm-3 and that of water vapour is 0.0006 g cm-3 then the volume occupied by waetr molecules in 1 L of steam at this tem perature is :
Options
A.6 cm3
B.60 cm3
C.0.6 cm3
D.+ 6.8 cm3
Solution
Mass of 1000 mL steam = 1000 × 0.0006
= 0.6g
∴ Volume of liquid water =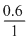 = 0.6 cm3
= 0.6 cm3
= 0.6g
∴ Volume of liquid water =
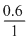 = 0.6 cm3
= 0.6 cm3 Create a free account to view solution
View Solution FreeMore Gaseous State Questions
The density of neon will be highest at :...The coordination number of a metal crystallizing in a hexagonal close packed structure is :...The volume of a gas increases by a factor of 2 while the pressure decreases by a factor of 3. Given that the number of m...The compressibility factor for an ideal gas is ?...2.5 L of a sample of a gas at 27oC and 1 bar pressure is compressed to a volume of 500 mL keeping the temperature consta...