Coordination CompoundHard
Question
The pair in which both species have same megnetic moment (spin only value) is :
Options
A.[Cr(H2O)6]2+, [CoCl4]2-
B.[Cr(H2O)6]2+, [Fe(H2O)6]2-
C.[Mn(H2O)6]2+, [Cr(H2O)6]2+
D.[CoCl4]2-, [Fe(H2O)6]2+
Solution
[Cr(H2O)6]2+. Here Cr is in Cr2+ form
Cr2+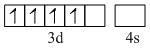
Fe2+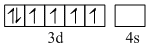
In [Fe(H2O)]2+, Fe is in Fe2+ form. Both will have 4 unpaired electrons.
Cr2+
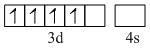
Fe2+
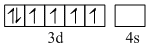
In [Fe(H2O)]2+, Fe is in Fe2+ form. Both will have 4 unpaired electrons.
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
A complex containing cobalt (III) ion is associated with two diaminopropane molecules and two chloride ions forming an o...Select the correct statement from the following ....The magnetic moment of a salt containing Zn2+ ion is :...Which of the following name is impossible ?...Select incorrect statement(s) for [Cu(CN)4]3–, [Cd(CN)4]2– and [Cu(NH3)4]2+ complex ion....