Coordination CompoundHard
Question
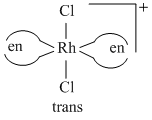
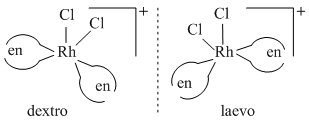
A complex containing cobalt (III) ion is associated with two diaminopropane molecules and two chloride ions forming an octahedral geometry. The complex ion will show :
Options
A.ligand isomerism
B.geometrical isomerism
C.optical isomerism
D.ionization isomerism
Solution
Diaminopropane can exist as 1,2,- diaminopropane (pn) and 1,3-diaminopropane (tn.) So exhibits ligand isomerism.


Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
The pair in which both species have same megnetic moment (spin only value) is :...In Fe(CO)5, the Fe - C bond possesses...Following Sidwick′s rule of EAN, Co(CO)x will be :...Malachite is an ore of :...Which one of the following is an outer orbital complex and exhibits paramagnetic behaviour ?...