ThermochemistryHard
Question
For a phase change,
H2O (l)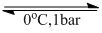 H2O(s)
H2O(s)
H2O (l)
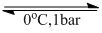 H2O(s)
H2O(s)Options
A.ᐃ = 0G
B.ᐃS = 0
C.ᐃH = 0
D.ᐃU = 0
More Thermochemistry Questions
The heat of reaction for : C10H8(s) + 12O2(g) → 10CO2(g) + 4H2O(l) at constant volume is 1228.2 kcal at 25oC. The ...CsF(S) → Cs+(aq) + F-(aq) ᐃH = - 40 kJ mol- If lattic energy of CsF is 750 kJ mol- . Then the summation of h...Which of the following reaction(s) is/are endothermic?...A + BA gives iodoform test. ′B′ is heated with excess of H-I. then ′C′ is obtained A and C are :...The heat evolved in the combustion of glucose C6H12O6 is −680 kcal/mol. The mass of CO2 produced, when 170 kcal of heat ...