ThermochemistryHard
Question
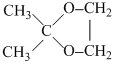
A gives iodoform test. ′B′ is heated with excess of H-I. then ′C′ is obtained A and C are :-
Options
A.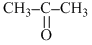 , CH3-CH2-I
, CH3-CH2-I
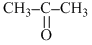 , CH3-CH2-I
, CH3-CH2-IB.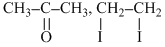
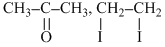
C.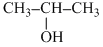 , CH3-CH2-I
, CH3-CH2-I
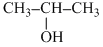 , CH3-CH2-I
, CH3-CH2-ID.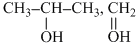
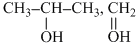
More Thermochemistry Questions
The polymerization of ethylene to linear polyethylene is represented by the reactionnCH2 = CH2→ (−CH2−CH2−)nwhere n has ...50.0 mL of 0.10 M HCl is mixed with 50.0 mL of 0.10 M NaOH. The solution′s temperature rises by 3.0oC. Calculate t...The enthalpies of combustion of carbon and carbon monoxide are -393.5 and -283 kJ mol-1 respectively. The enthalpy of fo...NH3(g) + 3Cl2(g) → NCl3(g) + 3HCl(g) ; ᐃH1N2(g) + 3H2(g) → 2NH3(g) ; ᐃH2H2(g) + Cl2(g) → 2...From the following thermochemical equations, find out the bond dissociation enthalpy of CH3−H bond.CH3I(g) → CH3(g) + I(...