ThermodynamicsHard
Question
During isothermal expansion of the mole of an ideal gas from 10 atm at 273K, the workdone is [gas constant = 2]:
Options
A.-895.8 cal
B.- 1172.6 cal
C.- 1257.43 cal
D.- 1499.6cal
Solution
Work done in expansion of gas
= - 2.303 nRT log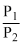
= -1257.43cal.
= - 2.303 nRT log
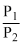
= -1257.43cal.
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
The following reaction is performed at 298 K. 2NO(g) + O2(g) ⇋ 2NO2(g) The standard free energy of formation of NO...Standard entropy of X2, Y2 and XY2 are 60, 40 and 50 JK-1mol-1, respectively. For the reaction,, ᐃH = - 30 kJ, to ...An ideal gas expands in volume from 1 × 10-3 m3 to 1 × 10-2 m3 at 300 K against a constant pressure of 1 ×...One mole of a non-ideal gas undergoes a change of state (2.0 atm, 3.0 L, 95 K) with a change in internal energy, (ᐃ...The entropy change involved in the isothermal reversible expansion of 2 moles of an ideal gas from a volume of 10 dm3 to...