ThermodynamicsHard
Question
The following reaction is performed at 298 K. 2NO(g) + O2(g) ⇋ 2NO2(g) The standard free energy of formation of NO(g) is 86.6 kJ/mol at 298 K. What is the standard free energy of formation of NO2(g) at 298 K ? (Kp = 1.6 × 1012)
Options
A.86600 - 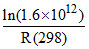
B.0.5[2 × 86,600 − R(298) ln (1.6 × 1012)]
C.R(298) ln(1.6 × 1012) − 86600
D.86600 + R (298) ln(1.6 × 1012)
Solution
ᐃGorxn = 2ᐃGof (NO2 ) − 2ᐃGof (NO)
ᐃGof (NO2) =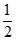 [ᐃGorxn + 2ᐃGof (NO)]
[ᐃGorxn + 2ᐃGof (NO)]
ᐃGof(NO2) = 0.5[2 × 86600 − R(298) ln (1.6 × 1012)]
ᐃGof (NO2) =
ᐃGof(NO2) = 0.5[2 × 86600 − R(298) ln (1.6 × 1012)]
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
Heat of combustion of ethanol at constant pressure and at temperature TK (= 298 K) is found to be - q J mol-1. Hence, he...Calculate the equilibrium constant for the reaction :PCl5 ⇋ PCl3 + Cl2 (g) (g) (g)at 400K if ᐃHo = 69418.86 ...For the reaction of the one mole of zinc dust with one mole of sulphuric acid in a bombcalorimeter, ᐃU and w corre...The correct expressions for an adiabatic process are...Internal energy does not include:...