ThermodynamicsHard
Question
Standard entropy of X2, Y2 and XY2 are 60, 40 and 50 JK-1mol-1, respectively. For the reaction,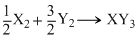 , ᐃH = - 30 kJ, to be at equilibrium, the temperature will be
, ᐃH = - 30 kJ, to be at equilibrium, the temperature will be
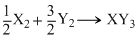 , ᐃH = - 30 kJ, to be at equilibrium, the temperature will be
, ᐃH = - 30 kJ, to be at equilibrium, the temperature will beOptions
A.1250 K
B.500 K
C.750 K
D.1000 K
Solution
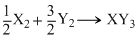
ᐃSreaction = 50 -
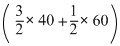 = - 40 j mol-1
= - 40 j mol-1 ᐃG = ᐃH - TH - TᐃS
at equilibrium ᐃG = 0
ᐃH = TᐃS
30 × 103 = T × 40
⇒ T = 750 K
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
Which of the following graphs between pressure 'P' versus volume 'V' represent the maximum work done?...The efficiency of the reversible heat engine is ηr and that of irreversible heat engine is ηi. Which of the following re...The minimum work which must be done to compress 16 g of oxygen isothermally at 300 K from a pressure of 1.01325 × 103 N/...The dissolution of NH4Cl in water is endothermic even through NH4Cl dissolves in water spontaneously. Which one of the f...The work involved (w) in an isothermal expansion of n moles of an ideal gas from an initial pressure of ‘P’ atm to final...