ThermodynamicsHard
Question
The entropy change involved in the isothermal reversible expansion of 2 moles of an ideal gas from a volume of 10 dm3 to a volume of 100 dm3 at 27oC is :
Options
A.35.8 J mol-1K-1
B.32.3 J mol-1K-1
C.42.3 J mol-1K-1
D.38.3 J mol-1K-1
Solution
For an ideal gas, for isothermal reversible process,
ᐃS = 2.303nR log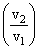
= ᐃS = 2.303 × 2 × 8.314 log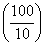
= 38.3 J mol-1 k-1
ᐃS = 2.303nR log
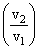
= ᐃS = 2.303 × 2 × 8.314 log
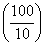
= 38.3 J mol-1 k-1
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
At 300 K, the reactions which have the following values of thermodynamic parameters, occur spontaneously....For a reaction, A ⇌ P, the plots of [A] and [P] with time at temperatures T1 and T2 are given below. If T2 > T1...The ΔG in the process of melting of ice at −15°C and 1 atm, is...An ideal gas is expanded irreversibly from 5 L to 10 L against a constant external pressure of 1 bar. The value of heat ...6 mole of an ideal gas expand isothermal and reversibly from a volume of 1 liter to avolume of 10 liters at 27oC. What i...