Chemical Kinetics and Nuclear ChemistryHard
Question
Which of the following electrolyte will have maximum flocculation value for Fe (OH)3 sol ?
Options
A.NaCl
B.Na2S
C.(NH4)3 PO4
D.K2SO4
Solution
Flocculating value ∝ 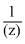 , where z = valency of coagulating ion.
, where z = valency of coagulating ion.
Since, (OH)3 is a positively charged sol so it will be coagulated by nagative ions con sidering the valencies of various nagative ions i.e., Cl-, S2-, PO43- and SO42-, we find that the valency of Cl- is least so it will have maximum flocculation value i.e NaCl.
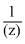 , where z = valency of coagulating ion.
, where z = valency of coagulating ion.Since, (OH)3 is a positively charged sol so it will be coagulated by nagative ions con sidering the valencies of various nagative ions i.e., Cl-, S2-, PO43- and SO42-, we find that the valency of Cl- is least so it will have maximum flocculation value i.e NaCl.
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Kyoto Protocol was endorsed at :...T50 of first - order rection is 10 min. Starting with 10 mol-1 rate after 20 min is...For the first-order reaction (C = C0e-k1t) and Tav = K1-1. After two average lives concentration of the reactant is redu...The hypothetical reaction : 2A + B → C + D is catalyzed by E as indicated in the possible mechanism below -Step-I ...For a given reaction A → Product, rate is 1 × 10-4 M s-1 when [A] = 0.01 M and rate is 1.41 × 10-4 M s-1...