Chemical Kinetics and Nuclear ChemistryHard
Question
For the first-order reaction (C = C0e-k1t) and Tav = K1-1. After two average lives concentration of the reactant is reduced to :
Options
A.25%
B.75%
C.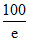 %
%
D.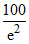 %
%
Solution
Given C = C0e-k1t
tav =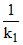
Given t = 2 tav =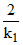
C = C0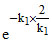
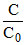 × 100 =
× 100 = 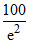
tav =
Given t = 2 tav =
C = C0
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
If $I$ is the intensity of absorbed light and C is the concentration of AB for the photochemical processAB + hν → AB*, t...The rate constant of a reaction is given by k = 2.1 × 1010 exp(-2700/RT). It means that...For a zero-order reaction,...For a second order reaction plots are made for vs time for the reaction, 2A → Product. Pick up the correct sentenc...For a complex (multistep) reaction,...