Chemical Kinetics and Nuclear ChemistryHard
Question
T50 of first - order rection is 10 min. Starting with 10 mol-1 rate after 20 min is
Options
A.0.0693 mol-1 min-1
B.0.0693 × 2.5 mol L-1 min-1
C.0.0693 × 5 mol L-2 min-1
D.0.0693 × 10 mol L-1 min-1
Solution
Initial concentration = 10 mol-1
∴ Conc. after 20 min(two half lives) = 2.5 mol-1
Now, k =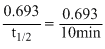
or 0.0693 min-1
∴ rate = k × [reactant]
= 0.0693 × 2.5 mol L-1 min-1
∴ Conc. after 20 min(two half lives) = 2.5 mol-1
Now, k =
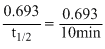
or 0.0693 min-1
∴ rate = k × [reactant]
= 0.0693 × 2.5 mol L-1 min-1
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
In a consecutive reaction system $A\overset{\quad E_{1}\quad}{\rightarrow}B\overset{\quad E_{2}\quad}{\rightarrow}C$ whe...The physical adsorption of gases on the solid surface is due to:...Select correct statement (s) :...The rate of chemical reaction is directly proportional to :...The rate constant of first order reaction is 3 × 10-6 per second. the initial concentration is 0.10 M.The intial ra...