Chemical Kinetics and Nuclear ChemistryHard
Question
Which of the following graphs represent relation between initial concentration of reactants and half-life for third order reaction ?
Options
A.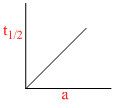

B.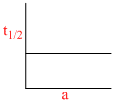

C.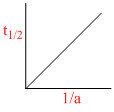

D.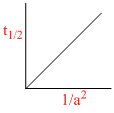

Solution
Graph (d) represents graph between t1/2 and initial concentration for 3rd order reaction:
(a) Zero order reaction
(b) 1 st order reaction
(c) 2nd order reaction
(a) Zero order reaction
(b) 1 st order reaction
(c) 2nd order reaction
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Which of the following electrolyte will have maximum flocculation value for Fe (OH)3 sol ?...For the non-stoichiometre reaction 2A + B → C + D, the following kinetic data were obtained in three separate expe...Which of the following statement is incorrect about half-life period :...When excess toluene-α-d (C6H5CH2D) was photo chemically mono chlorinated at 80°C with 0.1 mole of chlorine, there was ob...For the first-order reaction t99% = x × t90%. The value of ‘x’ will be...