Chemical Kinetics and Nuclear ChemistryHard
Question
Which of the following statement is true for the reaction H2 + Br2 → 2HBr. The rate law is 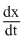 = k[H2][Br2]1/2 :
= k[H2][Br2]1/2 :
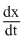 = k[H2][Br2]1/2 :
= k[H2][Br2]1/2 :Options
A.order of reaction is 1.5
B.molecularity of the reaction is 2
C.by increasing the concentration of Br2 four times the rate of reaction is doubled
D.All the above are correct.
Solution
All the statements are correct,
Order of reaction = 1 +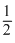 = 1.5
= 1.5
molecularity = 1 + 1 = 2
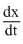 α [Br2]1/2
α [Br2]1/2
So. reaction rate will be doubled if concenration of Br2 is increased by 4 times.
Order of reaction = 1 +
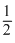 = 1.5
= 1.5molecularity = 1 + 1 = 2
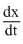 α [Br2]1/2
α [Br2]1/2So. reaction rate will be doubled if concenration of Br2 is increased by 4 times.
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Which of the following statement(s) is/are correct ?...Rate law of the reaction A → Product is, rate = k[A]. Graphically it is represented as Hence, rate constant is :...Under the same reaction conditions, initial concentration of 1.386 mol dm-3 of a substance becomes half in 40 seconds an...3A → B + CIt would be a zero order reaction when :...T50 of first - order rection is 10 min. Starting with 10 mol-1 rate after 20 min is...