Chemical Kinetics and Nuclear ChemistryHard
Question
Under the same reaction conditions, initial concentration of 1.386 mol dm-3 of a substance becomes half in 40 seconds and 20 seconds through first order and zero order kinetics, respectively. Ratio 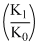 of the rate constants for first order (k1) and zero order (k0) of the reactions is
of the rate constants for first order (k1) and zero order (k0) of the reactions is
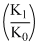 of the rate constants for first order (k1) and zero order (k0) of the reactions is
of the rate constants for first order (k1) and zero order (k0) of the reactions isOptions
A.0.5 mol-1 dm3
B.1.0 mol dm-3
C.1.5 mol dm-3
D.2.0 mol-1 dm3
Topic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
For the first order reaction :...The correct increasing order of basic strength for the following compounds is :...For the reversible reaction A$\rightleftharpoons$B; ΔH = −2 kcal, the pre-exponential factors are same for the forward a...On increasing the temperature, the half-life of reactions...In terms of polar character, which of the following order is correct :-...
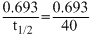
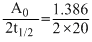
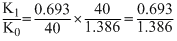 = 0.5 mol-1litre
= 0.5 mol-1litre