Ionic EquilibriumHard
Question
When 10 mL of 0.1 M acetic acid (pKa = 5.0), is titrated against 10mL of 0.1 M ammonia solution (pKa = 5.0), the equivalence point occurs at pH :
Options
A.5.0
B.6.0
C.7.0
D.9.0
Solution
CH3COOH + NH4OH → CH3COONH4 + H2O for salt of weak base & weak acid
pH = -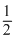 [log Ka + logKw - log Kb]
[log Ka + logKw - log Kb]
= -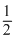 [-pKa + kogKw - logKb]
[-pKa + kogKw - logKb]
= -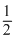 [-5 + log10-14 + 5]
[-5 + log10-14 + 5]
= 7
pH = -
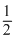 [log Ka + logKw - log Kb]
[log Ka + logKw - log Kb]= -
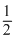 [-pKa + kogKw - logKb]
[-pKa + kogKw - logKb]= -
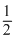 [-5 + log10-14 + 5]
[-5 + log10-14 + 5]= 7
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
The solubility of BaSO4 in water 2.42 × 103 gL–1 at 298 K. The value of solubility product (Ksp) will be (Gi...The pH of 10-8 M solution of HCl in water is :...Which group of animals belong to the same phylum?...Common hydrolysis so products of XeF2 & XeF4 are :-...The following graph represents the titration of pH vs volume Find out the best possible statement from the graph shown....