Chemical EquilibriumHard
Question
Steam reacts with iron at high temperature to give hydrogen gas and Fe3O4(s). The corrent expression for the equilibrium constant is
Options
A.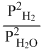
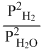
B.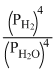
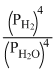
C.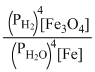
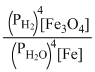
D.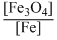
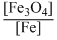
Solution
3Fe(s) + 4H2O(steam) ⇋ Fe3O4(s) + 4H2(g)
KP =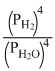
only gaseous products and reactants.
KP =
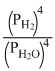
only gaseous products and reactants.
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
The first and second dissociation constants of an acid H2A are 1.0 × 10-5 and 5.0 × 10-10 respectively.The ove...What can you say about the existance of A if the potential energy diagram for the reaction A → B looks like...The correct relationship between free energy change in a reaction and the corresponding equilibrium constant K is...For a given exothermic reaction, Kp and K′p are the equilibrium constants at temperatures T1 and T2, respectively....Carbon monoxide in water gas reacts with steam according to the following reaction.CO(g) + H2O(g) $\rightleftharpoons$CO...