Chemical EquilibriumHard
Question
The first and second dissociation constants of an acid H2A are 1.0 × 10-5 and 5.0 × 10-10 respectively.
The overall dissociation constant of the acid will be
The overall dissociation constant of the acid will be
Options
A.5.0 × 10-5
B.5.0 × 1015
C.5.0 × 10-15
D.0.0 × 105
Solution
H2A ⇋ HA- + H+ K1 =  .....(1)
.....(1)
HA- ⇋ H+ + A2- K2 =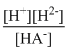 .....(2)
.....(2)
For the reaction
H2A ⇋ 2H+ + A2-
K =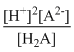 = K1 × K2
= K1 × K2
= 1 × 10-5 × 5 × 10-10
= 5 × 10-5
Hence, (3) is correct.
 .....(1)
.....(1)HA- ⇋ H+ + A2- K2 =
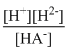 .....(2)
.....(2) For the reaction
H2A ⇋ 2H+ + A2-
K =
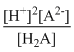 = K1 × K2
= K1 × K2 = 1 × 10-5 × 5 × 10-10
= 5 × 10-5
Hence, (3) is correct.
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
Given the following reaction at equilibrium N2(g) + 3H2(g) ⇋ 2NH3(g). Some inert gas at constant pressure is added...For a reversible reaction A + B$\rightleftharpoons$C, if the concentrations of the reactants are doubled at a definite t...For dissociation of a gas N2O5 as N2O5(g) $\rightleftharpoons$2NO2(g) + ½ O2(g). The reaction is performed at constant t...The position of equilibrium will shift in the given direction by the addition of inert gas at constant pressure in which...For the following gases equilibrium, N2O4 (g) ⇋ 2NO2 (g)Kp is found to be equal to Kc. This is attained when :...