AIPMT | 2014Chemical EquilibriumHard
Question
For a given exothermic reaction, Kp and K′p are the equilibrium constants at temperatures T1 and T2, respectively. Assuming that heat of reaction is constant in temperature range between T1 and T2, it is readily observed that
Options
A.Kp > K′p
B.Kp < K′p
C.Kp = K′p
D.Kp = 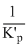
More Chemical Equilibrium Questions
When CO2(g) is dissolved in water, then the following equilibrium is establishedCO2(aq) + 2H2O(l) $\rightleftharpoons$H3...For the following mechanism, P + Q PQ R at equilibrium is :[k represents rate constant]...The value of Kp for the reaction, 2H2O(g) + 2Cl2(g) ⇋ 4HCl (g) + O2 (g)is 0.03 atm at 427oC, when the partial pres...The value of ᐃH for the reactionX2(g) + 4Y2(g) ⇋ 2XY4(g) is less than zero. Formation of XY4(g) will be favo...1 mol of N2 is mixed with 3 mol of H2 in a litre container. If 50% of N2 is converted into ammonia by the reaction N2 + ...