Ionic EquilibriumHard
Question
The pH of solution containing 0.10 M sodium acetate and 0.03 M acetic acid is
(pKa for CH3COOH = 4.57)
(pKa for CH3COOH = 4.57)
Options
A.0.09
B.6.09
C.5.09
D.7.09
Solution
According to henderson equation, pH of acidic buffer is given as
pH = pKa + log
= 4.57 + log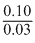
= 4.57 + [log 10 - log 3]
= 4.57 + log 3.33
= 4.57 = 0.52 = 5.09
pH = pKa + log

= 4.57 + log
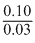
= 4.57 + [log 10 - log 3]
= 4.57 + log 3.33
= 4.57 = 0.52 = 5.09
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Which of the following will occur if a 0.1 M solution of a weak acid is diluted to 0.01 M at constant temperature?...A saturated solution of silver benzoate (AgOCOC6H5) has pH of 8.6. Ka for benzoic acid is 5.0 × 10–5. The value of Ksp f...25 ml of a solution of barrium hydroxide on titration with a 0.1 molar solution of hydrochloric acid gave a litre value ...The pH of 4.0 × 10−4 M-HNO3 solution is (log 2 = 0.3)...Which solution will have pH closer to 1.0:...