Ionic EquilibriumHard
Question
pKa value of four acids are given below. The strongest acid is :
Options
A.4.0
B.3.5
C.2.5
D.2
Solution
Strength of acid ∝ Ka
or ∝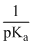
Hence acid with lostest pKa value (2.0) will be strongest.
or ∝
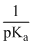
Hence acid with lostest pKa value (2.0) will be strongest.
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
A weak acid (or base) is titrated against a strong base (or acid), volume v of strong base (or acid) is plotted against ...100 mL of 0.02 M benzoic acid (pKa = 4.2) is titrated using 0.02 M NaOH. pH after 50 mL and 100 mL of NaOH have been add...The pH at which water is maximum dissociated at 25o C, is...Which of the following statements is correct for a solution saturated with AgCl and AgBr if their solubilities in moles ...Which would decrease the pH of 25 ml of a 0.01 M solution of hydrochloric acid? The addition of...