Ionic EquilibriumHard
Question
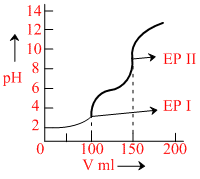
A weak acid (or base) is titrated against a strong base (or acid), volume v of strong base (or acid) is plotted against pH of the solution (as shown in figure). The weak electrolyte (i.e. acid or base) could be

Options
A.Na2CO3
B.Na2C2O4
C.H2C2O4
D.CH2(COOH)2
Solution
Given graph is plotted for the titration of weak diprotic acid with base.
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Let the colour of the indicator HIn (colourless) will be visible only when its ionised form (pink) is 25% or more in a s...Two buffer solutions, A and B, each made with acetic acid and sodium acetate differ in their pH by one unit, A has salt ...The lassaigne′s extract is boiled with conc. HNO3 while testing for the hologens by doing so it :-...The solubility products of Al(OH)3 and Zn(OH)2 are 8.5 × 10-23 and 1.8 × 10-14 respectively. If NH4OH is added...The pH at the equivalence point when a solution of 0.01 M-CH3COOH is titrated with a solution of 0.01 M-NaOH, is (pKa of...