Ionic EquilibriumHard
Question
100 mL of 0.02 M benzoic acid (pKa = 4.2) is titrated using 0.02 M NaOH. pH after 50 mL and 100 mL of NaOH have been added are
Options
A.3.50, 7
B.4.2, 7
C.4.2, 8.1
D.4.2, 8.25
Solution
C6H5COOH + OH- → C6H5COO- + H2O
t = 0 2 1
teq 1 - 1
pH = pKa = 4.2
C6H5COOH + OH- C6H5COO- + H2O
t=0 2 2
teq - - 2
[C6H5COO-] =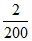 = 0.01 M
= 0.01 M
∴ pH = 7 + pKa +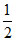 log C = 7 +
log C = 7 + 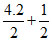 log (0.01) = 8.1
log (0.01) = 8.1
t = 0 2 1
teq 1 - 1
pH = pKa = 4.2
C6H5COOH + OH- C6H5COO- + H2O
t=0 2 2
teq - - 2
[C6H5COO-] =
∴ pH = 7 + pKa +
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
An amount of 0.1millimole of CdSO4 is present in 10 ml acid solution of 0.08 M-HCl. Now H2S is passed to precipitate all...The pH of 0. 1 M solution of the following salts increases in the order :...Solubility product constant (Ksp) of salts of types MX, MX2 and M3X at temperature, T are 4.0 × 10−8, 3.2 × 10−14 and 2....Arsenic (III) sulphide form a sol with negative charge. Which of the following ionic substances will have lowest floceul...The degree of dissociation of water at 25°C is 1.8 × 10–7% and the density is 1.0 g cm–3. The ionic product for water is...