Ionic EquilibriumHard
Question
One mole of CH3COOH and one mole of C2H5OH reacts to produce 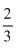 mol of CH3COOC2H2. The equilibrium constant is:
mol of CH3COOC2H2. The equilibrium constant is:
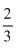 mol of CH3COOC2H2. The equilibrium constant is:
mol of CH3COOC2H2. The equilibrium constant is:Options
A.2
B.+2
C.-4
D.+4
Solution
CH3COOH + C2H5OH → CH3COOC2H5 + H2O
At start 1 mole 1 mole 0 0
Atequb. 1 -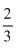 1 -
1 - 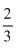
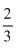
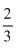
Let the total volume = V L
∴ [CH3OOH] =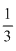 V mol/L
V mol/L
[C2H5OH =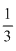 V mol/L
V mol/L
[CH3COOC25] =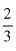 V mol/L
V mol/L
[H2O] =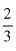 V mol/L
V mol/L
∴ K =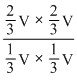 = 4
= 4
At start 1 mole 1 mole 0 0
Atequb. 1 -
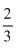 1 -
1 - 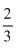
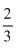
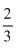
Let the total volume = V L
∴ [CH3OOH] =
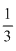 V mol/L
V mol/L[C2H5OH =
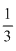 V mol/L
V mol/L[CH3COOC25] =
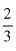 V mol/L
V mol/L[H2O] =
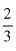 V mol/L
V mol/L∴ K =
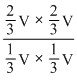 = 4
= 4Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
pH of a mixture containing 0.10 M X- and 0.20 M HX is: [pKb (X-) = 4]...Which of the following solutions when added to 1L of a 0.01 M CH3COOH solution will cause no change in the degree of dis...pH of a 0.1 M monobasic acid is found to be 2. Hence its osmotic pressure at a given temperature TK is :-...An alkali is titrated against an acid with methyl orange as indicator, which of the following is a correct combination ?...The equilibrium carbonate ion concentration after equal volumes of 0.7 M-Na2CO3 and 0.7 M-HCl solutions are mixed, is (K...