Ionic EquilibriumHard
Question
pH of a 0.1 M monobasic acid is found to be 2. Hence its osmotic pressure at a given temperature TK is :-
Options
A.0.1 RT
B.0.11 RT
C.1.1 RT
D.0.01 RT
Solution
Given pH = 2 ∴ H+ = 10-2
H+ = C α → α =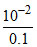 = 0.1
= 0.1
Now α =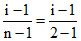
or 0.1 = i - 1 or i = 1.1
π = i CRT
= 1.1 × 0.1 × RT
= 0.11 RT
H+ = C α → α =
Now α =
or 0.1 = i - 1 or i = 1.1
π = i CRT
= 1.1 × 0.1 × RT
= 0.11 RT
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
The best explanation for the solubility of MnS in dilute HCl is that :...Solubility of BaF2 in a solution of Ba(NO3)2 will be represented by the concentration term...Pure ammonia is placed in a vessel at a temperature where its dissociation constant (α) is appreciable. At equilibr...A volume of 50 ml of a solution which is 0.05 M in the acid HA (pKa = 3.80) and 0.08 M in HB (pKa = 8.20) is titrated wi...A solution is prepared in which 0.1 mole each of HCl, CH3COOH and CHCl2COOH is present in a litre. If the ionization con...