Ionic EquilibriumHard
Question
pH of a mixture containing 0.10 M X- and 0.20 M HX is: [pKb (X-) = 4]
Options
A.4 + log 2
B.4 - log 2
C.10 + log 2
D.10 - log 2
Solution
pOH = pKb + log 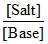 = 4 + log
= 4 + log 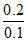 ∴ pOH = 4 + log 2 and pH = 10 - log 2
∴ pOH = 4 + log 2 and pH = 10 - log 2
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Which of following can act as buffer ?...Which of the following statement(s) is/are correct?...What mass of NaOH should be dissolved in sufficient water to get 20 m3 of an aqueous solution of pH 7.3 at 25o C?...Which is/are correct statements : (a) When 100 ml of 0.1 M NaCN solution is titrated with 0.1 M HCl solution the variati...What is the solubility of MnS in pure water, assuming hydrolysis of S2– ions?Ksp of MnS = 2.5 × 10–10, Ka1 = 1 × 10–7 an...