Ionic EquilibriumHard
Question
Which of the following solutions when added to 1L of a 0.01 M CH3COOH solution will cause no change in the degree of dissociation of CH3COOH and pH of the solution ? Ka = 1.6 × 10-5 for CH3COOH?
Options
A.0.6 mM HCOOH (Ka = 8 × 10-4)
B.0.1 M CH3COONa
C.0.4 mM HCl
D.0.01 M CH3COOH
Solution
(A) In 0.6 m M HCOOH solution
Ka =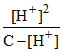 ⇒ 8 × 10-4 =
⇒ 8 × 10-4 = 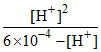
[H+] = 4 × 10-4 M.
So solution in (A), (C) & (D) are ISOHYDRIC.
Ka =
[H+] = 4 × 10-4 M.
So solution in (A), (C) & (D) are ISOHYDRIC.
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
pH value of which of the following is not equal to one :...The pH of an acid buffer can be raised by 2 units by...The relation pKa = 14 – pKb is true for which of the following pair(s)?...Which would decrease the pH of 25 ml of a 0.01 M solution of hydrochloric acid? The addition of...A solution has initially 0.1 M-HCOOH and 0.2 M-HCN. The value of Ka for HCOOH = 2.56 × 10−5, Ka of HCN = 9.6 × 10−10. Th...