ElectrochemistryHard
Question
The electrode potential of electrode
M(s) → Mn+ (aq) (2M) + ne-.
at 298 K is E1. When temperature is doubled and concentration is made half, then the electrode potential becomes E2. Which of the following represents the correct relationship between E1 and E2 ?
M(s) → Mn+ (aq) (2M) + ne-.
at 298 K is E1. When temperature is doubled and concentration is made half, then the electrode potential becomes E2. Which of the following represents the correct relationship between E1 and E2 ?
Options
A.E1 > E2
B.E1 < E2
C.E1 = E2
D.Cann′t be predicted
Solution
E1 = Eo - 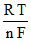 ln 2
ln 2
E2 = Eo -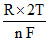 ln1 = Eo
ln1 = Eo
∴ E2 > E1
E2 = Eo -
∴ E2 > E1
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
100 mL of buffer of 1 M NH3(aq) and 1 M NH4+(aq) are placed in two comparments of a voltaic cell separately. A current o...Four successive members of the first row transition elements are listed below with atomic numbers. Which one of them is ...In a galvanic cell, the salt bridge...At 298K the standard free energy of formation of H2O(l) is -237.20 kJ/mole while ᐃGo of its ionisation into H+ ion...The reaction Zn(s) + 2AgCl(g) → ZnCl2(aq) + 2Ag(s) occurs in the cell Zn |ZnCl2 (1M solution), AgCl(s) | Ag. The n...