ElectrochemistryHard
Question
An aqueous solution of X is added slowly to an aqueous solution of Y as shown in List-I. The variation in conductivity of these reactions is given in List-II. Match List-I with List-II and select the correct answer using the code given below the lists :
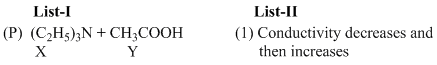
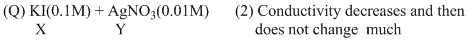
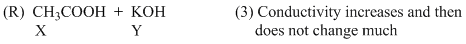
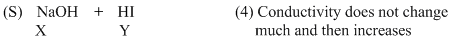
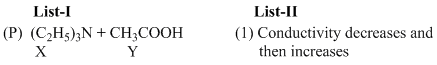
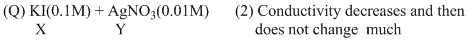
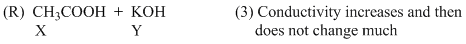
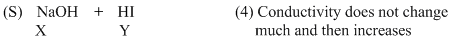
Options
A.P → 3, Q → 4, R → 2, S → 1
B.P → 4, Q → 3, R → 2, S → 1
C.P → 2, Q → 3, R → 4, S → 1
D.P → 1, Q → 4, R → 3, S → 2
Solution
(A) For weak base v/s weak acid titration upto equivalence point conductivity increases because weakelectrolyte (W.A.) is converted to strong electrolyte (salt) but after it weak base does not dissociateappreciably
(B) For KI + AgNO3 → AgI(ppt.) + KNO3
upto equivalence point number of ions remain same but after it increases
(C) For weak acid v/s strong base titration upto equivalence point OH- ion is replaced by CH3COO-so conductivity decreases but after it weak acid does not dissociate appreciably.
(D) For strong base v/s strong acid titration upto equivalence point H+ ion is replaced by Na+ so conductivitydecreases but after it increases due to increase in number of ions.
(B) For KI + AgNO3 → AgI(ppt.) + KNO3
upto equivalence point number of ions remain same but after it increases
(C) For weak acid v/s strong base titration upto equivalence point OH- ion is replaced by CH3COO-so conductivity decreases but after it weak acid does not dissociate appreciably.
(D) For strong base v/s strong acid titration upto equivalence point H+ ion is replaced by Na+ so conductivitydecreases but after it increases due to increase in number of ions.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Using same quantity of current, which among Na, Mg and Al is deposited more (by mass) during electrolysis of their molte...In a galvanic cell...The ionic mobility of alkali metal ions in aqueous solution is maximum for...The following electrochemical cell has been set up: Pt(s)|Fe3+, Fe2+ (a = 1)||Ce4+, Ce3+ (a = 1)|Pt(s); E°(Fe3+|Fe2+) = ...In an analytical determination of arsenic, a solution containing arsenious acid, H3AsO3, KI and a small amount of starch...